Laboratory Information Management Systems (LIMS)
What is a Laboratory Information Management System (LIMS)?
Ensure data quality from sample collection through project completion.
Maintain a robust compliance posture and proactive risk management across your laboratory operations.
Optimize your labs performance through real-time data, actionable insights and trusted KPIs.
What Capabilities Make a
Best-in-Class LIMS?
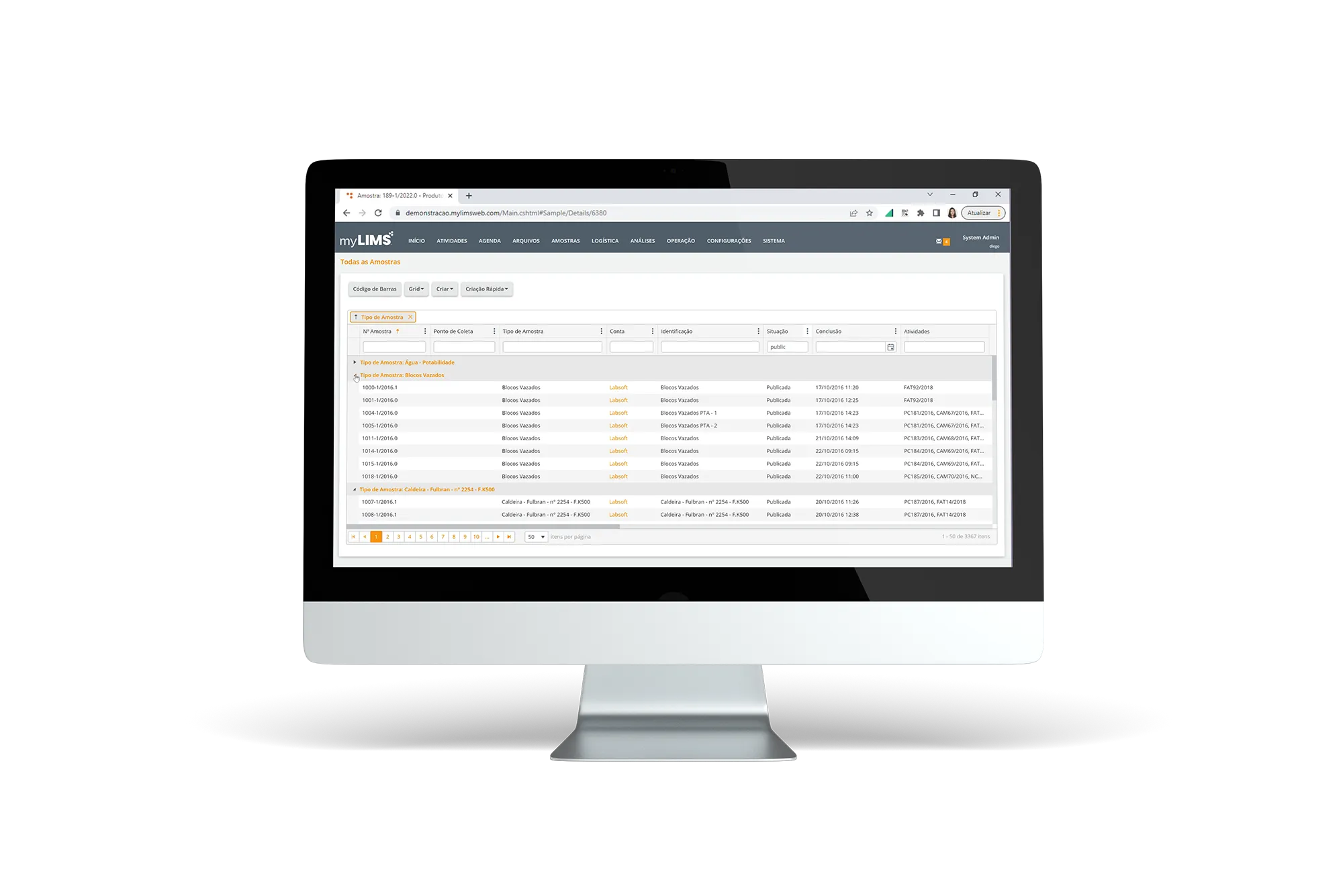
LIMS software streamlines lab operations with features enhancing efficiency, accuracy, and compliance. It automates workflows and sample tracking to optimize processes and reduce errors. LIMS ensures high data integrity through verification and regulatory compliance functions, and maintains transparent audit trails for accountability. Integration with instruments and APIs facilitates smooth data flow, while robust security features protect sensitive information. The system is scalable, allowing labs to expand without losing performance, and introduces cost-efficiency metrics along with advanced analytics for better decision-making. LIMS is essential for modern labs aiming to improve efficiency and support growth.

Next, match potential LIMS solutions to your requirements. Dive into product demos, review vendor certifications, assess their track record and reputation, and consider feedback from existing clients. Don’t forget to evaluate the technology itself to ensure it aligns with your lab’s expertise and future aspirations.
Discover the steps The Caribbean Industrial Research Institute (CARIRI) took in selecting a LIMS.
Here’s some additional tips to help you confidently choose a LIMS that not only addresses your lab’s current challenges but also evolves with your needs over time.
Ready to get started?
Next, match potential LIMS solutions to your requirements. Dive into product demos, review vendor certifications, assess their track record and reputation, and consider feedback from existing clients. Don’t forget to evaluate the technology itself to ensure it aligns with your lab’s expertise and future aspirations.
Discover the steps The Caribbean Industrial Research Institute (CARIRI) took in selecting a LIMS.
Here’s some additional tips to help you confidently choose a LIMS that not only addresses your lab’s current challenges but also evolves with your needs over time.
Ready to get started?
Product Specification Management
The key to product quality is adherence to published specification limits. The Confience myLIMS Specification Manager allows the definition of multiple limit ranges, customer specification limits, statistical quality control limits as well as textual limits and issues warnings whenever conformance is violated.
Sample Login and Test Assignment
A variety of options are available to rapidly create samples and assign descriptive information both manually and automatically via interfaces and schedules. Rules for the assignment of tests and associated limits can be based on the process step to ensure that the right tests are performed at the right time.
Result Entry Options
A variety of results entry options are available to accommodate your lab’s preferences. Manual entry can be performed by sample, by test, by worksheet or by crosstab. In addition, results can also be entered using mobile devices such as tablets and smart phones. Of course, the most efficient means of results entry is by instrument interface and if Excel is your preference, Confience myLIMS can be linked directly to well-defined spreadsheets.
Dashboard Web Parts
User-specific, interactive dashboard web parts are configured to provide actionable information that is specific to each person’s job function. Dynamic workflow lists and interactive graphs are powerful tools that minimize user’s need to navigate through the software because the information required to guide workflow is always available from their dashboard.
Management of projects, customers, pricing, costs, priorities, turnaround times, inventory and other organizational information are inherent to Confience myLIMS. A portal that allows requestors to submit their own samples, retrieve the status of their requests and have reports automatically issued when the lab has completed the work request is also included to eliminate time and error.
Imagine a lab where customers submit their own samples, barcode labels are printed, prioritized worklists are created, instrument data flows into the system and reports are automatically generated with little or no manual interaction. In addition, laboratory inventory is managed, and instrument calibration and maintenance schedules are self-generated. Cost and KPI reports are ready at your fingertips. This is the power of Confience myLIMS.
Additional myLIMS features that benefit Analytical and Commercial Testing labs include:
Customer Portal
A web portal allows your customers to create their own requests and samples while avoiding inefficiencies that are often experienced by eliminating duplication of effort and transcription errors. The portal also allows customers to monitor the progress of work, access validated and/or approved data and retrieve their reports without disrupting the laboratory staff with phone calls and emails.
Workflow Management
The status of lab requests, samples, tests and schedules are automatically updated within interactive dashboard web parts to allow the throughput to be continuously monitored. With little or no effort, you will identify and circumvent problems before they occur. In addition, barcodes can be utilized to positively identify and track samples as they are processed.
Automated Reports and Emails
Reports can be sent automatically based on status change or timing to ensure that information flows to the right people at the right time. This means that COAs and invoices can be issued to customers upon final approval, notifications & warnings can be sent to responsible staff members via email or text, labels can be printed, and management reports can be sent without any time-consuming, manual effort.
Instrument Interfaces
Data generated from instruments can be imported directly into Confience myLIMS to save time and transcription error. This includes QA/QC data that will automatically generate SQC reports for instruments and associated standards.
Quotation and Invoicing
Basic pricing for each Test and for pre-defined Test Groups can be assigned within Confience myLIMS. In addition, customer-specific pricing is also available if required. Site-specific prioritization levels can also be implemented to automatically calculate turnaround times (compensated for weekends and holidays) and adjust the prices of requests, samples and/or individual tests. This information can be used to generate quotations and invoices. Invoice data can be passed to your accounting system to provide a seamless flow of information from beginning to end.
R&D labs need a system that allows them to design experiments, record both structured and flexible data, and keep everything organized for quick access and reporting. They also require tools to search records, attach files, link web pages, cross-reference projects, and customize features for their specific needs.
Confience myLIMS addresses these challenges with a highly configurable LIMS and built-in Lab Notebook features that allow R&D labs to integrate files, perform free-text searches, and access past data with ease—streamlining collaboration and eliminating redundant tasks.
Confience myLIMS offers flexible features tailored for R&D labs across all industries. Users can easily submit samples online, track request statuses, and receive automated reports once testing is complete.
Plus, Confience myLIMS offers powerful tools to enhance efficiency and productivity, such as:
File Attachment & Free Text Searching
Files of any type (images, movies, PDFs, Word, Excel, etc.) can be easily attached to Confience myLIMS records to associate information for quick retrieval. Confience myLIMS includes a query tool (Smart Search) that allows rapid retrieval of information using words and phrases by simultaneously searching the database records and attachments to locate records that would be irretrievable from notebooks or network files.
Configurable Page Designs
Pages and the underlying tables that manage data can be designed and modified throughout the life of the system to allow you to dynamically change the system as your needs change without costly code modification.
Product Formulation Management
Experimental formulations can be recorded within Confience myLIMS which records the individual ingredients, supplier, material lot, and amounts and serves as a repository for future reference. In addition to retaining the ingredients, the process steps can also be recorded. The material produced by the formulation can be cross-referenced with test results that are obtained to allow formulations to be optimized to produce a product with the desired properties.
Project Management
A Project Management tool is included within Confience myLIMS to organize work for easy retrieval and reporting. A security feature enables you to identify project team members and securely restrict access to data associated with those projects to project team members only.

How can a LIMS help me achieve and maintain industry compliance?
Regulatory compliance isn't just a requirement, it's essential for maintaining quality, ensuring safety, and avoiding costly setbacks. A LIMS simplifies compliance by automating data management, ensuring traceability, and keeping your lab audit ready at all times.
Accreditation also provides internal and external customers with assurances regarding the quality, reliability and reproducibility of testing data.
LIMS are an essential part of achieving and maintaining these accreditations and can provide the backbone to any laboratory’s QMS.
Confience myLIMS streamlines sample tracking from start to finish—login to disposal—capturing every critical step needed for reliable, defendable results. It securely documents the entire process to ensure:
- Various versions of test methods, dates of activation and retirement
- Training record management for analysts and re-certification dates
- Full on-screen audit trail on results
- Complete chain of custody with date/time stamps
- Chemical inventory, lot numbers, expiration dates and storage information
- Instrument maintenance, calibration and repair logs
- Control charts and internal laboratory standards tracking
- Corrective and Preventive Actions (CAPA)
- Subcontracting sample tracking
- Reporting templates and Electronic Data Deliverables (EDDs)
LIMS are instrumental in supporting GLP certifications, which are critical for ensuring the quality and reliability of scientific data in research and regulatory environments. Key elements for GLP certifications include Standard Operating Procedures (SOPs), quality assurance, documentation, reporting, personnel, facility and equipment management, and corrective and preventive actions (CAPA)s. LIMS software delivers essential data to support compliance in these areas.
Confience myLIMS streamlines and automates lab processes, ensuring that data is captured accurately and consistently, minimizing human errors and increasing reproducibility. Confience myLIMS provides comprehensive audit trails, controlled data access, and secure data management, all of which are fundamental components of GLP standards.
By maintaining detailed records of every modification and transaction with a robust audit trail, Confience myLIMS helps laboratories comply with the rigorous documentation requirements of GLP.
Additionally, the ability of Confience myLIMS to integrate with other lab instruments and enterprise systems enables a seamless flow of data across platforms, enhancing traceability and accountability. This level of organization and control not only supports compliance with GLP but also enhances the overall efficiency and effectiveness of laboratory operations.
Confience myLIMS plays a pivotal role in supporting GMP in various ways, including:
- Streamlining Sample Management
- Standard Operating Procedures (SOPs)
- Documentation and Record Keeping
- Quality Control and Validation
- Personnel Training and Qualifications
- Facility and Equipment
- Improving Batch Release Processes
- Audit Readiness
- Scaling Operations
Why Choose a Software as a Service (SaaS) LIMS?
Confience myLIMS offers a fast, cost-effective way to implement and manage your laboratory information management system. Hosted securely in the cloud, our solution ensures high speed connectivity, robust security, and seamless performance. With a SaaS model, you'll receive continuous updates, expert management, and the flexibility of a predictable monthly subscription - so you can focus on what matters most: running your lab efficiently.

Reduced Time to Benefit
Using the Confience myLIMS SaaS (Software as a Service) model reduces the time spent on installation and configuration and can reduce internal IT issues that often interfere with software deployment.
Lower Costs
Confience myLIMS SaaS provides beneficial cost savings through its low monthly pricing and frees up your budget for other needs. Additionally, SaaS software is treated as an expense which has great tax advantages.
Scalability and Integration
Confience myLIMS SaaS resides in a fully scalable cloud environment. This means that the system can start small (in size and cost) and grow by increasing the server’s memory, disk space and CPU as you grow without any interruption to laboratory operation.
Discover how switching to a SaaS LIMS solution has revolutionized Fairfax County Department of Public Works and Environmental Services’ approach to managing lab operations, data, and compliance.










